Written by

14th December 2021 – Value-based care has been prevalent in the US for many years, and more recently several other countries have started to roll out healthcare reform plans that also focus on improving the quality of healthcare provision, whilst increasing efficiency of the care provided. This is subsequently affecting product choice, with healthcare providers assessing product features more extensively, to ensure the level of care provided is optimised. In the respiratory and anaesthesia care markets, solutions that enable the highest level of patient safety are prioritised, which in turn is driving demand for connected solutions with advanced product functionality.
The spread of value-based care
The innovation seen in the US has been looked on by other countries, especially those with a large proportion of publicly funded healthcare and who have seen increased strain on healthcare provision. Although countries outside of the US have not employed guidelines as extensive as the HITECH Act, the shift toward reimbursement based on the value and quality of care provided is becoming more prevalent, with many countries employing clinical outcome incentives. This is driving the uptake of clinical care solutions that help to support hospitals in their bid to maintain the best possible return on their investment. Similar reforms have evolved and subsequently exist in other major countries, including China, Japan, and the UK.
Digitalisation
The need for information to support the data-driven insights utilised to establish value-based care has further driven the development of digital solutions. Digitalization of healthcare solutions enables the utilisation and sharing of patient information to ultimately improve care provision. With a focus on the continuum of patient care to reduce any risk of avoidable events, the priority to ensure patient data is collected throughout a patient’s care pathway is also increasing the need for digital solutions.
With more digital solutions being used, the demand for connected and interoperable solutions is also increasing. Developments to improve interoperability between devices have been evolving at a fast pace. This was primarily driven by the Meaningful Use Act in the United States which stipulated the requirement for medical devices to be interoperable, to not only connect to the electronic health record, but also to other clinical devices. Other countries closely followed with all major markets now focusing on connecting and integrating devices. HL7 is now the standard used to transfer clinical information between software and devices. However, additional standards are in development to further improve accuracy of the transition and utilisation of clinical information.
Impact on product choice – Anaesthesia and Ventilators
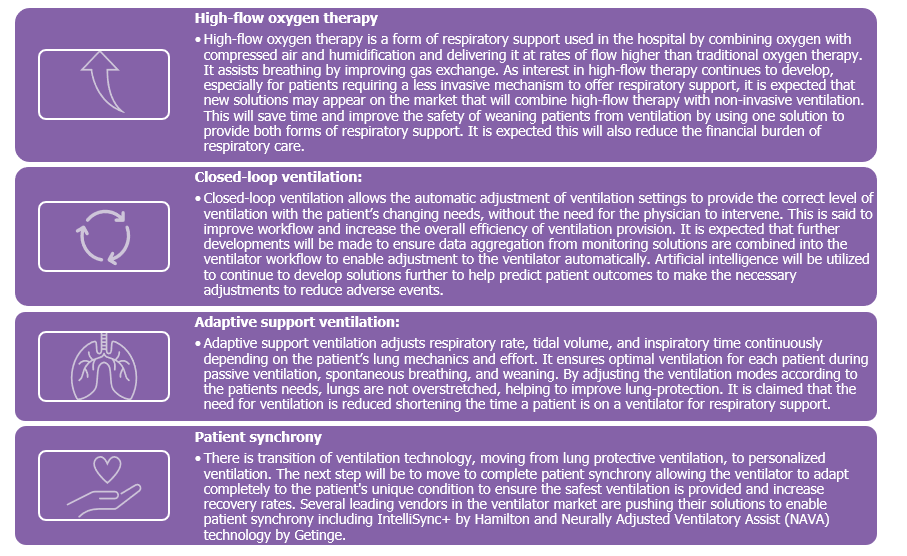
Connectivity of anaesthesia devices and ventilators is also now in greater demand, to enable the collection of necessary patient information to support data reporting. Mature regions are progressing to the next stage of data utilisation, assessing inefficiencies in patient care, and allowing adaptations earlier in the patient pathway. Product developments within the anaesthesia market that will assist in value-based healthcare provision include:

Product developments within the anaesthesia market that will assist in value-based healthcare provision include:
Closing remarks
With the standard of patient care now prioritised, there is a clear shift toward solutions that offer advanced functionality. Newer market entrants are also increasing the level of functionality of solutions at a more affordable price level. The premium segment of the anaesthesia market is projected with faster global annual revenue growth than all other segments from 2022 to 2025. For the respiratory care market, revenues for high-end critical care ventilators will remain the largest of all hospital-based ventilators through to 2025. The COVID-19 pandemic has further highlighted the need for solutions that can aid in patient provision, especially when healthcare resource is strained. Moving forward, the global emphasis in both markets is to advance to the highest possible level of care within the allocated budgets.
About the Reports
The recently published reports “Ventilators Report – 2021” and “Anaesthesia Devices Report – 2021” each provide a data-centric and global outlook of the respective markets. Each report blends primary data collected from in-depth interviews with healthcare professionals and technology vendors, to provide a balanced and objective view of the market.
About Signify Research
Signify Research is an independent supplier of market intelligence and consultancy to the global healthcare technology industry. Our major coverage areas are Healthcare IT, Medical Imaging and Digital Health. Our clients include technology vendors, healthcare providers and payers, management consultants and investors. Signify Research is headquartered in Cranfield, UK. To find out more: enquiries@signifyresearch.net, T: +44 (0) 1234 436 150, www.signifyresearch.net
More Information
To find out more:
E: enquiries@signifyresearch.net,
T: +44 (0) 1234 436 150