
Written by

23rd November 2022
Following attendance at BioTechX in Basel, Imogen Fitt from Signify Research shares her thoughts on the current landscape for using clinical data in Real-World Evidence Studies.
BioTechX was suggested as a good conference in which take the ‘pulse’ of the pharmaceutical world.
Three days of jam-packed talks and vendors discussions later, the show verified much of our recent research about the life sciences industry and its growing influence on a range of digital healthcare markets, especially surrounding ‘Big Data in Oncology, Image Analysis and Real-World Evidence’. A key show theme also focused on the use of clinical Real-World Data (cRWD) in research studies.
In the article below, we discuss what the conference’s discourse revealed about cRWD and it’s potential over the next few years.
What is Clinical Real-World Data?
First, when discussing the topic of cRWD, we must be careful to clearly define what is meant by the term.
Definitions for Real-World Data are broad, encompassing any health-related information collected outside the remits of a randomised clinical trial. In our most recent market study, Signify Research has developed the sub-definitions for use within our research, shown in Figure 1.
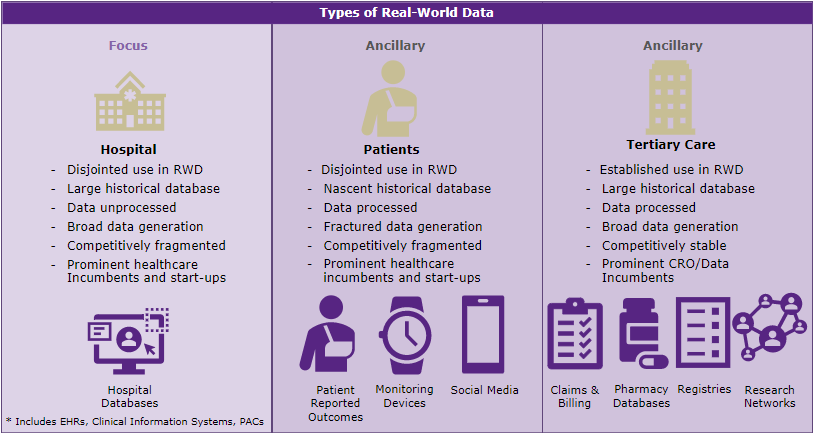
Figure 1: Topics explored during the scope of our study.
Whilst claims and registry data were discussed and utilised in abundance during presentations in Switzerland, more specific examples of large clinical datasets were notably scarce.
Instead, discussions revolved more around the specific challenges in utilising cRWD and how to overcome them.
One such example that stood out, revolved around EHR data and driving the use of FAIR principles in clinical data sources. FAIR principles dictate that data used for research should Findable, Accessible, Interoperable and Reusable, and whilst the points raised were well thought-out, an undiscussed elephant in the room remained obvious – there were no EHR health tech vendors, like Cerner, Epic, Allscripts at the conference or cited as partners.
Therefore, how could any progress be made without direct involvement from the architects of core clinical data software?
Instead, researchers and life science users would better benefit from looking at how these vendors choose to tackle the problem themselves.
A Platform-Centric Approach is the Future
Clinical data as it exists today is housed across multiple siloes of IT; disconnected structures as well as non-standardised terminology makes it hard even for providers to easily utilise.
The reason data remains so separated and unstructured is down to both a fragmented competitive environment and individual healthcare provider requirements for customisation. With no financial incentive, governmental push or clear benefit in standardising health data beyond very basic levels, providers aren’t investing in solving this challenge.
To try and counter this, multiple health tech vendors have developed platforms, collectively termed by Signify as ‘Data-Unification Platforms’, which are designed to circumvent this issue and aggregate data in an agnostic manner.
A few examples of such platforms available today are listed below:
- Cerner – HealtheIntent
- Epic – COSMOS
- Intersystems – HealthShare
It’s clear that the solution to utilising cRWD has been chosen by those experts who are most familiar with healthcare IT’s challenges. Healthcare data at its source will take years to transform into research-ready data formats, if it ever will. Instead, for cRWD to be utilised, clinical data must be aggregated and then transformed into a usable format.
But how should these platforms be developed to make the most out of opportunities available?
Users Driving Clinical RWD Use
There are currently multiple types of users for aggregated data cRWD, shown in Figure 2.
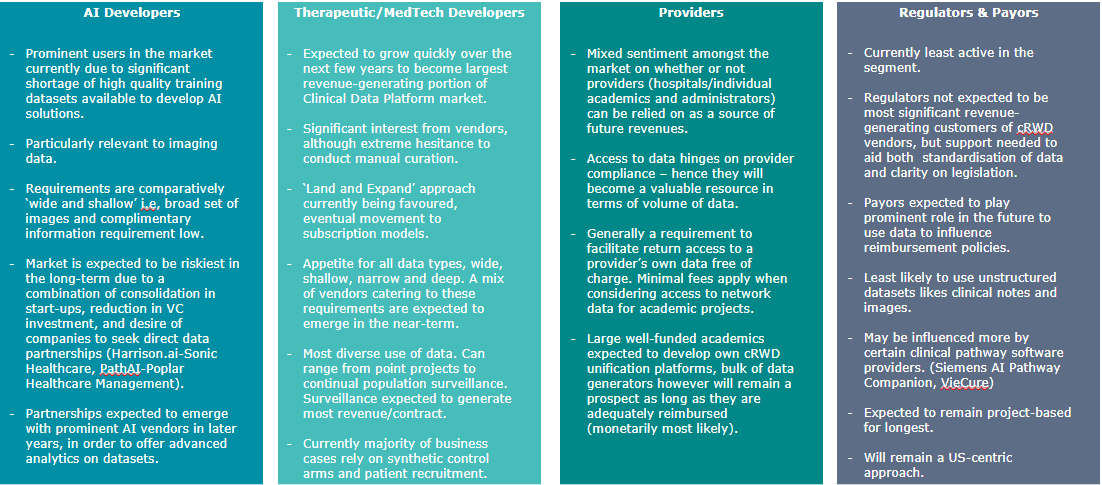
Figure 2: The main purchasers of clinical RWD.
Each type of prospective customer has unique requirements for that data and thus must be addressed individually. At the same time, for vendors the supply and acquisition of cRWD needs to be as cost-effective as possible to warrant desirable profit margins.
But cRWD is not usable in its native format, requiring sometimes extensive curation.
Within the cRWD market, the challenge of how-to approach curating datasets remains unanswered, with different vendors taking different approaches. Today, the topic sits on a spectrum, with some vendors preferring a ‘hands-off’ approach, leaving the majority of curation to individual users, whilst others are prepared to offer dedicated curation services, spending significant amounts of labour to completely solve the problem.
However curating data, especially unstructured data, into usable formats can quickly become time-consuming and costly, which is why most vendors are seeking to automate the process and reduce these costs as much as possible. The advent of advanced Optical Character Recognition, Natural Language Processing, Machine Learning, and Deep Learning technologies will be integral to this, although such technologies are relatively immature in the market today.
How will the market move forward?
cRWD use cases are so diverse that creating one unified, completely deidentified database fit for all purposes is unfeasible. There will always be a service element to these studies which tailor to individual requirements, and vendors must always be careful never to reveal too much information that may identify individuals.
Whilst individual markets will continue to address legislation independently, moving forward it’s clear that the chief barrier holding the market back today is arguably accessibility. Greater clarity and education is sorely needed to help users understand exactly what is possible, and at what price. Proper incentives should also be put in place in the market to help providers themselves engage with and contribute to these platforms, and finally the process of curation should be as simplified as is possible in order to reduce duplications in effort and increase data reusability.
Ultimately, data itself is like wheat in baking. Convert it to a platform, and it becomes flour with vast potential. Utilise it correctly, and you may have a souffle; incorrectly utilisation however it’s all too easy to end up a stick mess on the floor.
For more information about ‘Real-World Data – A Strategic Analysis – 2022’ please click here or email the report author directly at Imogen.Fitt@signifyresearch.net.
