Cerner, Epic, Allscripts and Meditech’s International EHR Endeavours


Alex Green
Published: June 19, 2019 In The News
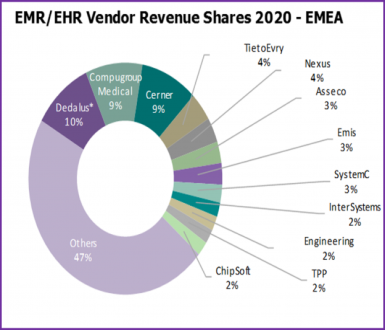
13th June 2019 – Written by Alex Green – Featured on Ramaon Healthcare – Late in 2018 signify Research provided an insight that outlined some of the key strategies US EHR vendors could employ to help maintain historical growth as their domestic core-EHR market started to saturate.
These strategies included:
- Diversifying product portfolios into related areas
- Expanding what is provided within ‚”standard‚” EHR offerings
- Geographic expansion
- Aggressive acquisition strategies that drive market share expansion
You may also like